Boiling Vaporization Melting and Condensation Are Described Best as
Learn the six changes of phase. To cause to become dissipated.
Boiling and Condensation When a liquid becomes a gas it is called boiling or vaporization.
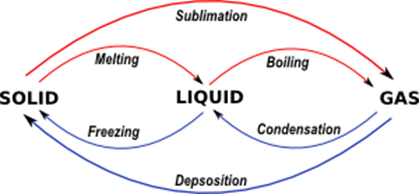
. Also Read- Difference Between Cell Membrane and Plasma Membrane. Fes Fet O vaporization condensation sublimation freezing 0 0 0 0 deposition melting. Condensation is when dew forms on grass in the morning.
Select the name of the physical state transition that is described in the following process. 6 The normal boiling point of water in an open container is 100C100C. Melting is the opposite of freezing.
Water is a liquid and iron is a solid at room. The temperature at which a substance changes from a solid to a. Opposite process of evaporation is condensation.
The point of condensation is the best description if the heating curve is reversed. Condensation is the transition from fuel to liquid as within the condensation of steam to liquid water. The point at which liquid changes to solid B.
THIS USER ASKED During which changes of state do atoms overcome the attractive forces between them. To convert as by the application of heat or by spraying into vapor. Condensation is the opposite process where water vapour is converted to tiny droplets of water.
Evaporation occurs before a liquid reaches its boiling point. Answer 1 of 2. The melting point for water is 0 degrees C 32 degrees F.
A water has a low heat of vaporization because it has weak intermolecular forces b heat of vaporization is the amount of energy needed to vaporize 1 g of a substance c the amount of energy released in condensation equals the amount of energy absorbed during vaporization. Freezing melting condensation vaporization sublimation and deposition. Condensation is a phase change regardless of the temperature.
Click on to see full reply Hereof whats the reverse of dissolving in science. Boiling point is when liquid turns into vapor. Condensation is the opposite since it is the turning point of vapor into liquid.
Which statement best describes the energy changes associated with evaporation and boiling. Select the single best answer. He watches as the flying birds land in neat little rows on.
Identify the different transition points between the phases of matter vaporization melting boiling freezing condensation sublimation Melting. Vaporization is the reverse from the liquid to the fuel state. Bboiling and deposition Ccondensation and melting Dvaporization and sublimation.
The point at which vapor changes to gas or liquidC. Assume the temperature is increased to 250 K and the pressure is increased to 100 bar. A phase is a distinctive form of a substance and matter.
Converting frost to water vapor melting boiling and evaporation in general are endothermic processes. The boiling point of a liquid depends on the inter-molecular fo. Select the single best answer.
5 A system where two processes such as vaporization and condensation occur simultaneously with equal rates such that the amount of substance in each phase is unchanged is described as _____. The point at which liquid changes to vapor D. Which of the following best describes the particles of a liquid.
Vaporization is when water boils and turns into steam. Condensation is the opposite of boiling. Freezing and condensation boiling and deposition condensation and melting vaporization and sublimation.
Which is not a change from liquid to gas state vaporization boiling condensation evaporation. Rahul sees a flock of birds. Which option best describes the term boilingA.
Condensation is the opposite process of evaporation. Amazon Luna launches with freebies for Prime subscribersAmazon Luna special offer for Prime membersTry Amazon Luna Now. Select the name of the physical state transition that is described in the following process.
Which statement describes the potential energy and the. The point at which solid changes to liquid. By definition evaporation is a process where water changes into vapour.
A condensation B deposition C solidification D vaporization 11. A substance is initially at the triple point. Melting is when those ice cubes melt.
What occurs when the rates of condensation and vaporization are the same. Plasma gas liquid and solid. A sample of water is boiling as heat is added at a constant rate.
Choose the following statements which best identify the differences between melting and freezing. The particles are far apart and moving fast. Both evaporation and boiling involve liquid changing to gas.
The heat associated with heating to the boiling point or cooling to the freezing point will be the same 50C the enthalpy of vaporization. Dude rock on THANKS. Advertisement New questions in Chemistry.
Which change of state is shown in the model. During which changes of state do atoms overcome the attractive forces between them. During condensation a gas loses energy and turns into a liquid.
The change in state from a solid to a liquid. But matter on Earth exists mostly in three distinct phases. A melting B deposition C sublimation D vaporization 12.
Changes of Phase There are four states of matter in the universe. What will the final phase of the substance be. What the vapor point is.
Select all the statements that correctly describe heat of vaporization. Which is NOT an endothermic process. To answer your question we need to know the liquid every element has a boiling point a point of vaporization and a point where it becomes a solid.
Condensation is the change of the physical state of matter from the gaseous phase into its liquid phase and is the reverse process of vaporization. W hich process decreases the potential energy of the particles of a sample. To destroy by or as if by converting into vapor a tank vaporized by a shell.
Deposition is one you may not know but this. When the opposite happens and a liquid turns into a solid it is called freezing. Gas liquid and solid.
Melting is from the strong to the liquid state and freezing from liquid to strong. It depends on the liquid you are boiling. Which of the following describes what happens.
Acondensation Bdeposition Cboiling. During condensation liquid water is formed from water vapour by cooling. Boiling by contrast is a rapid vaporization that occurs at or above the boiling temperature and at or below the liquids surface.
Boiling is a bulk phenomenon while evaporation is a surface phenomenon. This is called condensation. The correct answer is the first option.
These examples could be written as chemical reactions but are more generally considered to be endothermic or heat-absorbing processes.

What Is Vaporization Geeksforgeeks
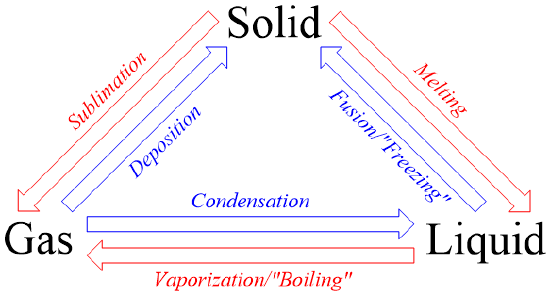
5 4 Phase Changes Chemistry Libretexts

Change Of State Of A Matter Melting Fusing Evaporation Condensation
Comments
Post a Comment